| Ch 10. Rankine Cycle | Multimedia Engineering Thermodynamics | ||||||
|
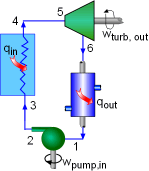
Rankine Cycle |
Reheat | Regeneration | Cogeneration | ||||
| Ideal Rankine Cycle | Case Intro | Theory | Case Solution |
| Chapter |
| 1. Basics |
| 2. Pure Substances |
| 3. First Law |
| 4. Energy Analysis |
| 5. Second Law |
| 6. Entropy |
| 7. Exergy Analysis |
| 8. Gas Power Cyc |
| 9. Brayton Cycle |
| 10. Rankine Cycle |
| Appendix |
| Basic Math |
| Units |
| Thermo Tables |
| eBooks |
| Dynamics |
| Fluids |
| Math |
| Mechanics |
| Statics |
| Thermodynamics |
| ©Kurt Gramoll |
|
|
||
| Introduction |
||
|
|
Neon, who works in a steam power plant, is asked to monitor the performance of a new system. The new system is running a Rankine cycle with water as the working fluid. What is known:
|
|
| Question |
||
VALIGN="TOP">
|
||
| Approach |
||
|
||