| Ch 6. Entropy | Multimedia Engineering Thermodynamics | ||||||
| Entropy | Tds Relations |
Entropy Change |
Isentropic Process |
Isentropic Efficiency |
Entropy Balance (1) |
Entropy Balance (2) |
Reversible Work |
| Entropy Change | Case Intro | Theory | Case Solution |
| Chapter |
| 1. Basics |
| 2. Pure Substances |
| 3. First Law |
| 4. Energy Analysis |
| 5. Second Law |
| 6. Entropy |
| 7. Exergy Analysis |
| 8. Gas Power Cyc |
| 9. Brayton Cycle |
| 10. Rankine Cycle |
| Appendix |
| Basic Math |
| Units |
| Thermo Tables |
| eBooks |
| Dynamics |
| Fluids |
| Math |
| Mechanics |
| Statics |
| Thermodynamics |
| ©Kurt Gramoll |
|
|
||
| Introduction |
||
|
|
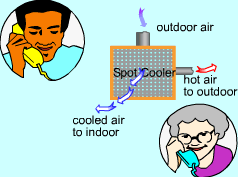
A spot cooler is a device which can produce hot and cold streams of air from a single stream of air at an intermediate temperature without any work or heat transfer. This product can be used for emergency cooling or short-term cooling. A seller called Kay and said his spot cooler can work at normal atmospheric pressure and cost nothing to cool the air. Can Kay trust the salesperson? What is known:
|
|
| Question |
||
|
|
Is the salesperson telling the truth? |
|
| Approach |
||
| If the product satisfies both the first and the second law of thermodynamics, it can work. To verify if it satisfies the first law, check the energy balance of the system. To verify if it satisfies the second law, check the entropy change of the entire process. | ||