| Ch 6. Entropy | Multimedia Engineering Thermodynamics | ||||||
| Entropy | Tds Relations |
Entropy Change |
Isentropic Process |
Isentropic Efficiency |
Entropy Balance (1) |
Entropy Balance (2) |
Reversible Work |
| Entropy | Case Intro | Theory | Case Solution |
| Chapter |
| 1. Basics |
| 2. Pure Substances |
| 3. First Law |
| 4. Energy Analysis |
| 5. Second Law |
| 6. Entropy |
| 7. Exergy Analysis |
| 8. Gas Power Cyc |
| 9. Brayton Cycle |
| 10. Rankine Cycle |
| Appendix |
| Basic Math |
| Units |
| Thermo Tables |
| eBooks |
| Dynamics |
| Fluids |
| Math |
| Mechanics |
| Statics |
| Thermodynamics |
| ©Kurt Gramoll |
|
|
||
| Introduction |
||
|
|
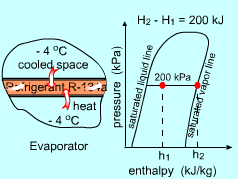
According to a recent new law, refrigeration systems are no longer allowed to use refrigerant R-12 due to environmental concerns. A cryogenic manufacturing facility has to choose a new refrigerant to replace R-12 used in its refrigeration systems. Refrigerant R-134a is one choice which satisfies the energy requirement. To further evaluate this new refrigerant, the entropy changes need to be determined. What is known:
|
|
| Question |
||
|
|
|
|
| Approach |
||
|
||